Over a half hour, a pale pink collection of stem cells immersed in a preservative dripped from a small bag through a line into Jessica Ceja’s arm.
They were her cells, reengineered in a lab in New Jersey with one goal in mind: a cure for her painful and destructive sickle cell disease.
This was her chance to “just be normal,” the 41-year-old said from her hospital bed at the University of Maryland Medical Center in Baltimore.
On Monday, Ceja was surrounded by her husband, doctors and several other members of her care team as she became the first patient in a Maryland hospital to get the new gene therapy.
The disease is not particularly common, with about 100,000 people across the country suffering. But it is particularly awful.
Gene mutations cause red blood cells, responsible for carrying oxygen around the body, to become misshapen and block blood flow. That’s given Ceja, a Silver Spring mother of three, nothing but intense bouts of pain, repeated trips to the emergency room and permanent damage to her body.
Read More
It’s most often diagnosed in people of African descent whose genes had mutated to resist malaria, but it also affects Hispanic people. Ceja was diagnosed as a toddler in El Salvador.
About 5,000 people in Maryland are living with the disease, the fifth-highest number of cases in the nation. That had sent the hospital’s doctors in search of more effective treatments.
Among the most promising is Lyfgenia, approved by the U.S. Food and Drug Administration in 2023 along with another gene therapy. The treatment involves harvesting a patient’s stem cells and genetically modifying them so that when they are reintroduced into a patient’s bone marrow, they produce new healthy red blood cells.
“No more pain episodes, strokes, damage to organs, necrosis of joints — all of it, hopefully,” said Dr. Jean Yared, a hematologist-oncologist treating Ceja.
The doctor took time to watch as four bags of Ceja’s cells were carefully removed from metal cassettes, checked and transported from the cryopreservation lab where they had been kept frozen in liquid nitrogen.
The manufacturer Genetix Biotherapeutics reports 90% of patients no longer have major episodes of pain up to 18 months after treatment.
The process is among an increasing number of therapies available or in the works, and doctors and patients will now have to weigh factors such as costs, prep times and potential drawbacks.
The medical center has already treated other patients with stem cell transplants, which are cheaper than the $2 million to $3 million for the gene therapies and can be done more quickly.
But patients’ bodies may reject transplanted cells and need long-term medications to minimize the risk. Transplants also require a perfectly matching sibling, which Ceja didn’t have.
Doctors have begun performing “half-match” transplants from other relatives, which could help expand the pool of donors.
So far, just over 100 people across the country have gotten Lyfgenia. The company’s CEO said last year that the “vast majority” of patients eligible hadn’t started treatment, well short of expectations for its use.
In addition to cost, that’s likely due to the labor involved. Ceja traveled four times for multiday stays since last May to collect enough stem cells to modify.
And when Ceja returned for the treatment last week, she withstood a blast of high-dose chemotherapy to clear diseased stem cells in preparation for the new cells. It left her extremely fatigued and subject to infection.
Dr. Edward Donnell Ivy, chief medical officer of the Sickle Cell Disease Association of America, said the process may not be for everyone.
Because it’s harsh and a huge time commitment, he said it’s best suited for relatively healthy, younger patients. It’s also too soon to call gene therapies a cure, as most patients will still have damage to their bodies and could still develop pain, he cautioned.
But the therapies are an important new tool, and more is needed to ensure patients and doctors know about them. Ivy said many patients now don’t have specialists or live near a center approved to offer gene therapies.
They rely on other treatments, as does Ivy, who has treated his own sickle cell disease for years with an older drug called hydroxyurea. That worked less well for Ceja.
Ivy said it reduced his pain, enabling him to go to medical school and become a doctor, though he’s suffered complications over time. Gene therapies should provide more relief to more people, and that is “something to celebrate,” he said.
“We need to make sure people are aware of all the therapies, including the new gene therapies,” he said. “And that access is not out of reach.”
The University of Maryland Medical Center is among dozens of sites now approved by manufacturers, said Yared, medical director of clinical operations in the hospital’s Marlene and Stewart Greenebaum Comprehensive Cancer Center.
Ceja first learned about the procedure when a 12-year-old patient, also from Maryland, was treated at Children’s National Hospital in Washington, D.C., where she works as a patient liaison.
In December, during her last round of stem cell collection, she let herself imagine a life free of pain as she lay tethered to a machine that filtered her cells from her blood.
Ceja said she has missed countless activities with her children, ages 5 to 18 years old. She leaned on her husband, Darwin, and her parents to carry more of the load. But mostly she limited herself, worried she’d get too tired or that a temperature change would trigger a pain crisis.
“It’s like having knives stabbing you repeatedly with no relief,” she said.
Dr. Jennie Law, a hematologist-oncologist also treating Ceja, said Ceja’s family support and her determination are reasons the hospital chose her as its first patient.
There are others also now getting the grueling treatment, and Law hopes over time it will be refined.
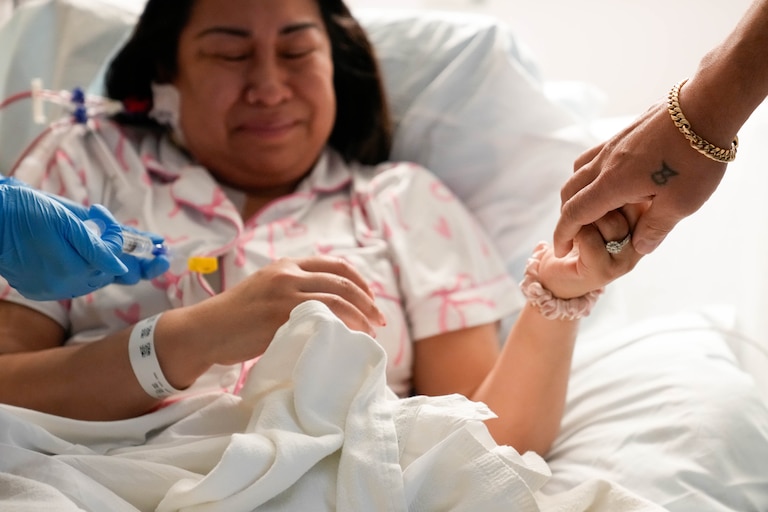
Ceja will be hospitalized for at least a month as her new cells generate, said Law, associate section chief for classical hematology and sickle cell disease in the cancer center.
Law also hopes to raise awareness of the therapy and close the disparities documented by the sickle cell association.
“Everyone should be able to be seen at a comprehensive sickle cell program to talk through these options,” she said. “There are a lot of reasons to hope.”
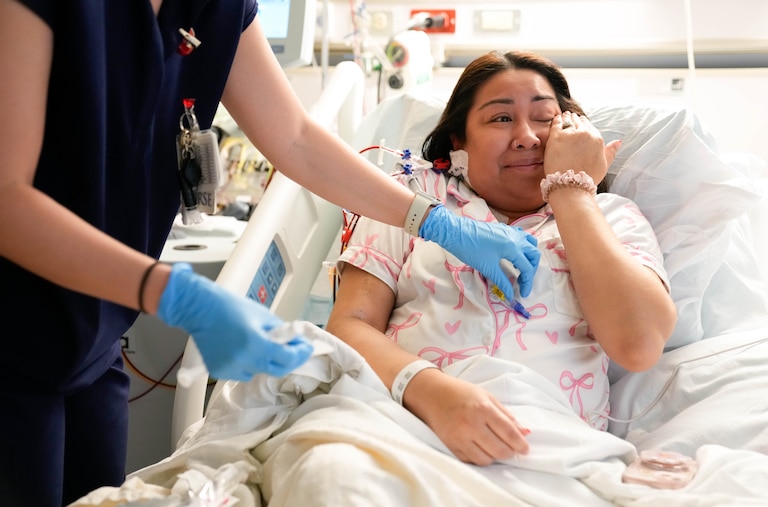
Four months after her last draw and a 11 months since she first came to the medical center, Ceja was back in a hospital bed draped in a Hello Kitty blanket with Darwin at her side.
There were tears, smiles and photos.
“I had no other option but to live life as is,” she said. “But here we are.”
For his part, Darwin has been keeping a wary eye on the calendar as his wife ages into her 40s. Sickle cell patients typically live 40 to 60 years. As her procedure this week approached, he said his main focus has been supporting her.
“God put me in her life for this reason, to give her strength, make her happy,” he said. “We hope this is the reward.”



Comments
Welcome to The Banner's subscriber-only commenting community. Please review our community guidelines.